- Validfor Demo is available now!
The AI-Native Agentic Digital Validation Platform
Validfor is an AI-native validation platform for life sciences, replacing static systems with intelligent, continuously compliant infrastructure.
Built for
GxP Environments
Designed for
Regulated Enterprises
Designed for
AI-Era Compliance
Backed by
Global VCs
Validation is Broken.
And Everyone Knows It.
Validation in Life Sciences is still driven by static documents, manual traceability, and outdated lifecycle tools. Teams spend months generating documentation that becomes obsolete the moment systems evolve.
Documentation
becomes the product
Teams focus on producing large volumes of documentation rather than enabling efficient, living validation processes.
Reactive
Compliance
Compliance activities happen after changes occur instead of being maintained continuously throughout the system lifecycle.
Validation can’t keep
up with AI Systems
Traditional validation approaches struggle to keep pace with dynamic, AI-driven systems that evolve constantly.
The result is slow releases, high costs, and
growing compliance risk.
From Validation Lifecycle Management
To AI-Native Validation Infrastructure
A new category is emerging.
Validation is no longer about managing documents.
It is about continuously understanding, validating, and adapting systems in real time.
Validfor is defining this new category.
From Static
Dynamic
Documentation
Intelligence
Lifecycle
Continuous Validation
A new category is emerging.
Validation is no longer about managing documents.
It is about continuously understanding, validating, and adapting systems in real time.
Validfor is defining this new category.
Meet
Validfor
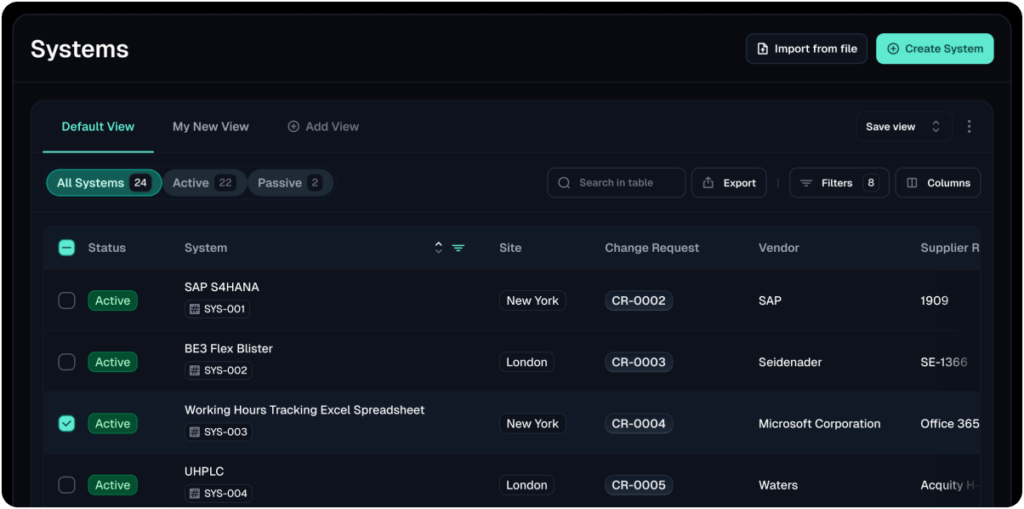
Validfor a Digital Validation Platform, gives life science teams a complete way to manage their validation lifecycle from one secure platform with a modular approach.
- Paperless
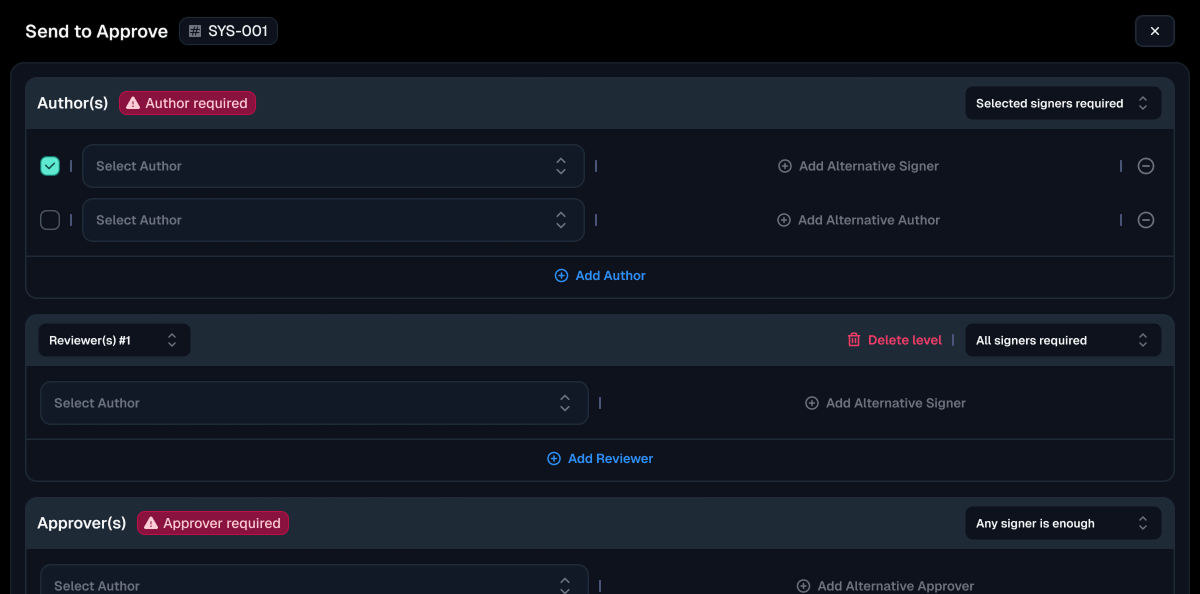
- Electronic Signature
- Electronic Record
Trust our secure platform to maintain top-tier data integrity, with interconnected records and real-time monitoring ensuring reliable, accurate data every time.
- Data Security & Distribution
- Modernised Audit Trails
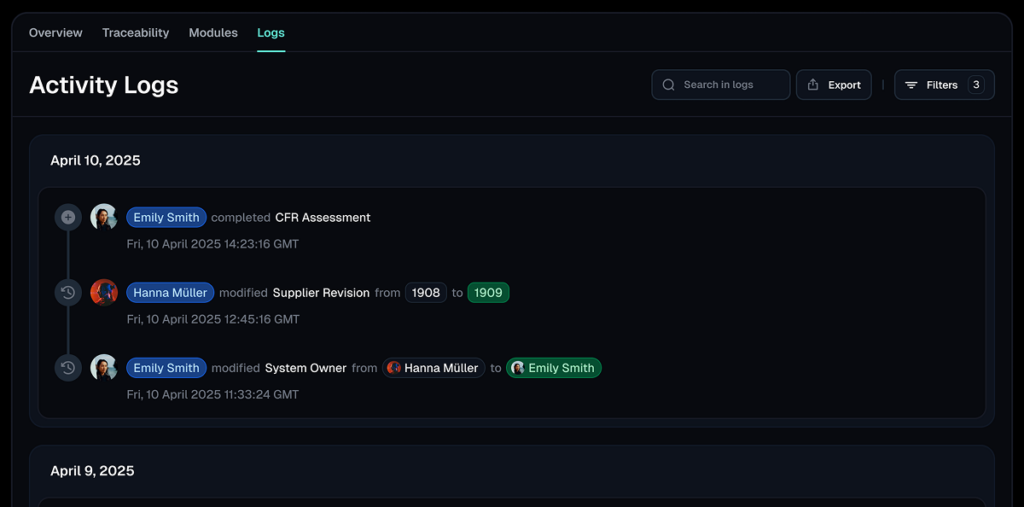
Validfor empowers comprehensive validation lifecycle management, simplifying every phase from implementation to retirement.
- Tailored Rules
- Real-Time & Dynamic
- Intelligent Linking
Validfor empowers your organization to lead eco-friendly validation by replacing outdated paper processes with a fully integrated digital platform.
- GreenShift
- DigiFlow
Validation becomes
a living system, not a static process.
Ready to move Beyond
Static Validation?
Digitalize validation your way, with the validation platform trusted by the world’s leading life sciences companies.
Powered by Agentic AI
Validfor uses a network of AI agents designed specifically for validation workflows in regulated environments.
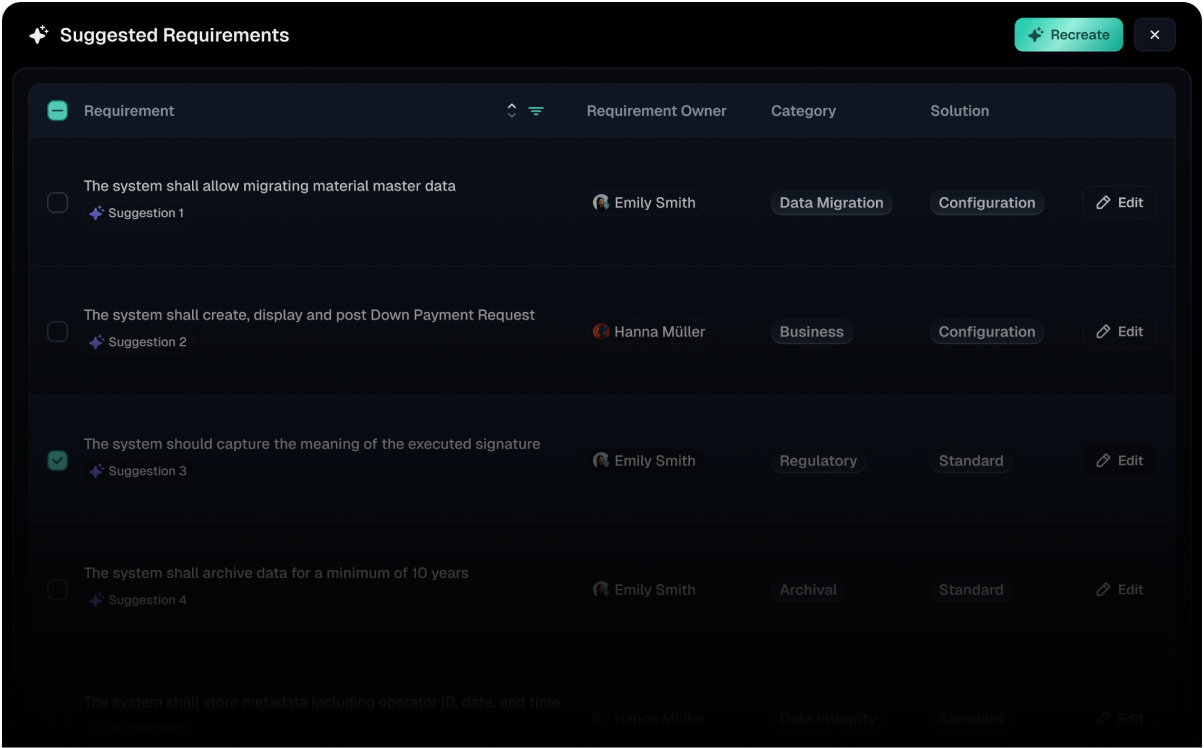
System
Understanding
Analyzes system requirements and detects changes automatically to keep validation aligned with evolving systems.
Automated
Validation Artifacts
Generates validation protocols, reports, and required documentation without manual effort.
Continuous
Compliance Monitoring
Maintains traceability and detects compliance gaps before they become audit risks.
Why Legacy Systems Cannot Compete?
Legacy VLM Tools
Document-Centric
Data Driven
Manual Workflows
Agent-Driven Workflows
Static Traceability
Dynamic Traceability
Reactive Compliance
Continuous Compliance
You cannot retrofit intelligence into
a system designed for documents.
What this means for you?
Heads of QA / Validation
Reduce validation timelines from months to days while maintaining audit readiness at all times.
Pharma Enterprises
Scale validation across complex, AI-driven systems without increasing operational burden.
Digital Transformation Teams
Enable innovation without being blocked by validation bottlenecks.
Built for
Real-World Validation Challenges
Validate faster. Stay compliant.
Computer System Validation (CSV)
Streamline validation for computer systems, guaranteeing accuracy and compliance.
Trust your AI decisions.
AI/ML System
Validation
Validate machine learning and AI systems to ensure reliability and regulatory compliance.
Compliance made simple.
GxP Compliance Workflows
Manage GxP processes efficiently while
maintaining full traceability and audit readiness.
Transform with confidence.
Digital Transformation Programs
Support your digital initiatives with structured validation and compliance frameworks.
Built for the Future of Pharma
Backed by global investors and designed for enterprise-scale deployment.
Trusted. Verified. Validfor.
Lukas B.
CSV Specialist
- 4.9/5
Game Changer for Validation
Validfor completely transformed how we handle system validations.
The platform is intuitive, automates repetitive tasks, and gives our team more time to focus on critical work.
Martina V.
QA Engineer
- 4.6/5
Reliable & Efficient
Since adopting Validfor, our validation workflows are faster and more reliable. Tracking and reporting compliance has never been easier.
Aisha P.
QS Specialist
- 4.8/5
Simplifying Compliance
Keeping up with regulations used to be stressful.
Validfor makes it straightforward and effortless, with clear dashboards and reminders that never let anything slip through
Sönke A.
VP Validation
- 5/5
Five Stars for Accuracy
Every step of our validation process is traceable and precise. Validfor ensures nothing is overlooked, giving us total confidence in our compliance.
Fenna V.
Consultant
- 4/5
Smart Automation
The AI-driven features are a lifesaver. Tasks that took hours are now done automatically, and we get actionable insights instantly.
Barnaby Q.
Compliance Director
- 4.2/5
Peace of Mind Guaranteed
Validfor provides full visibility across all systems. Knowing everything is monitored and validated correctly gives our team complete peace of mind.
We provide services
across the following countries
Netherlands
Italy
Belgium
Switzerland
Portugal
Korea
Canada
United States
Germany
Poland
India
Greece
France
Austria
Turkey
Spain
Great Britain
Ireland
Slovenia
Egypt
Bangladesh
Japan
North Macedonia
Saudi Arabia
Jordan
Finland
Puerto Rico
Iceland
Smarter insights,
bigger impact.
Computerized system validation is the backbone of safe,..
- Life Science, Validation Management
Data Integrity Policy for Pharmaceutical Industry is a set..
- Lifecycle, Validation Management
ALCOA principles are the five pillars, Attributable, Legible, Contemporaneous,..
- Life Science, Quality Assurance
Choose Validfor with confidence
Here are the compliance frameworks that Validfor's Trust Center follows which showcases our adherence to industry-standard security guidelines.
ISO 27001
A globally recognized standard for establishing, implementing, maintaining, and continually improving an information security management system (ISMS).
GDPR
The General Data Protection Regulation, a comprehensive data protection law in the EU, governs how organizations must protect personal data and privacy.
HIPAA
The Health Insurance Portability and Accountability Act, a U.S. law that mandates standards for protecting sensitive patient health information.
ISO 27701
ISO 27701 is an extension of ISO 27001 and ISO 27002 for privacy information management. It helps organizations comply with global privacy regulations by providing a framework for managing personal data.
ISO 27017
ISO 27017 is a standard providing guidelines for information security controls applicable to the provision and use of cloud services. It helps organizations ensure secure cloud services by implementing these best practices.
ISO 27018
ISO 27018 is a standard focusing on protecting personal data in the cloud. It sets out guidelines to ensure cloud service providers implement robust privacy and data security measures.
ISO 27005
Provides guidelines for information security risk management, which supports the concepts in ISO 27001 and is part of the broader ISO 27000 series.
ISO 22301
Specifies requirements for a management system to protect against, reduce the likelihood of, and ensure your organization recovers from disruptive incidents.
ISO 42001
ISO/IEC 42001 is the world’s first international AI management standard, providing a certifiable framework for developing and using AI systems responsibly and securely.
Frequently Asked
Questions
We have listed the most frequently asked questions and their answers about Validfor.
GENERAL
What is Validfor?
Validfor is a comprehensive, AI-powered platform designed to streamline and optimize the entire validation lifecycle for computerized systems in regulated industries, particularly the pharmaceutical sector.
Who should use Validfor?
Validfor is ideal for any organization in a regulated industry that needs to validate computerized systems, particularly those in the pharmaceutical, biotechnology, and medical device sectors. Our platform caters to various roles, including:
Validation professionals
Quality assurance teams
IT departments
System owners
Project managers
What makes Validfor different from other solutions?
- AI-powered assistance: Our platform leverages AI to provide intelligent guidance, automate tasks, and proactively identify risks, enhancing efficiency and accuracy.
- Comprehensive modules: Validfor offers a suite of integrated modules covering all aspects of validation, including change, test, deviation and periodic review modules.
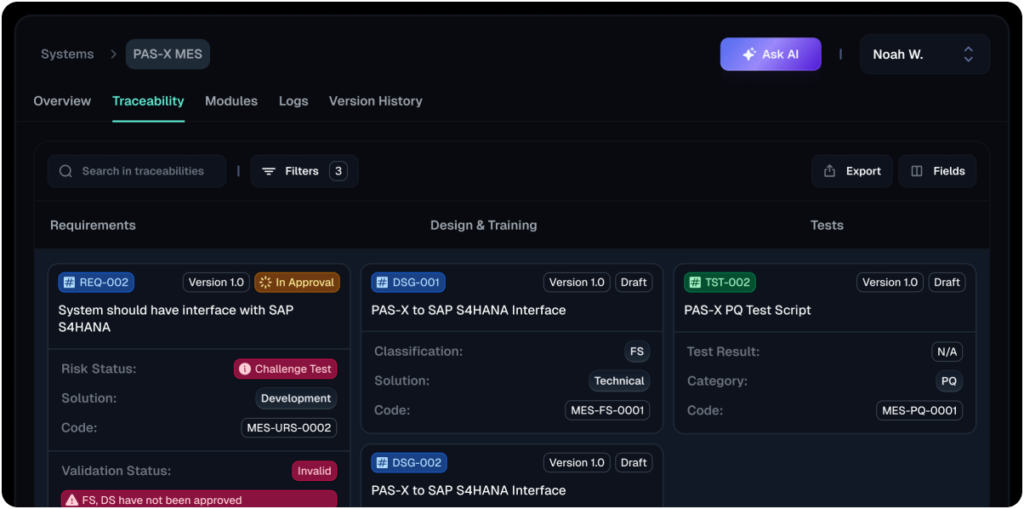
- Unmatched traceability:Our unique schematic view visualizes both primary and secondary linkages, providing unparalleled clarity and control over the impact of changes.
- Commitment to sustainability:Validfor promotes environmentally friendly validation practices through a digital-first approach, minimizing paper waste and reducing carbon footprint.
Compliance, Standards & Methodologies
What regulations and standards does Validfor support?
Validfor supports a wide range of regulations and standards, including GAMP 5, 21 CFR Part 11, and Annex 11. Our platform ensures data integrity, security, and auditability, enabling you to meet regulatory requirements and maintain compliance.
Can Validfor be used for both CSV and CSA?
Yes, Validfor supports both Computerized System Validation (CSV) and Computer Software Assurance (CSA), providing a unified platform for all your validation needs.
What types of computerized systems or equipment can be validated with Validfor?
Validfor can be used to validate a wide range of computerized systems, including:
- Enterprise Resource Planning (ERP) systems (e.g., SAP, Oracle)
- Manufacturing Execution Systems (MES) (e.g., PAS-X, POMSNet)
- Laboratory Information Management Systems (LIMS) (e.g., LabWare, LabVantage)
- Quality Management Systems (QMS) (e.g., TrackWise, MasterControl)
- Document Management Systems (DMS) (e.g., MasterControl, Veeva Vault)
- Building Management Systems (BMS) (e.g., Honeywell, Schneider)
- Production Equipment
- Laboratory Equipment
- Cleaning Validation
- And many more
Data Integrity & Security
How does Validfor ensure the integrity and security of data?
Validfor prioritizes data security and employs robust measures to protect your information, including:
- Data encryption: All data is encrypted both in transit and at rest.
- Access controls: Role-based access controls ensure that only authorized personnel can access sensitive data.
- Audit trails: Comprehensive audit trails track all user activities, providing a transparent record of data access and modifications.
- Regular backups: Data is regularly backed up to prevent data loss.
Secure infrastructure: Validfor is hosted on a secure and reliable infrastructure that meets industry best practices.
Product Implementation & Usage
How is Validfor implemented?
Validfor can be deployed cloud-based. Our team provides comprehensive implementation support, including configuration, data migration, and user training.
How does Validfor ensure that validation activities are completed on time and within budget?
Validfor provides tools for tracking timelines, resources, and costs associated with validation activities. It also offers alerts and notifications to help you stay on schedule and within budget.
What support does Validfor offer?
Validfor provides comprehensive support to our customers, including:
- Onboarding and training: We provide comprehensive onboarding and training to ensure your team can effectively utilize the platform.
- Technical support: Our technical support team is available to assist you with any questions or issues you may encounter.
- Documentation and resources: We provide extensive documentation and resources to help you get the most out of Validfor.
- Continuous improvement: We are committed to continuously improving Validfor based on customer feedback and industry best practices.
How can I get started with Validfor?
Contact us today to schedule a demo and learn how Validfor can transform your validation processes. Our team of experts will be happy to answer your questions and guide you through the implementation process.
Business & ROI
What is the typical ROI for using Validfor?
Validfor delivers a significant return on investment by increasing efficiency, reducing validation costs, minimizing risks, and improving compliance. Our customers typically see a return on their investment within the first year of implementation.
What is the pricing model for Validfor?
Validfor offers flexible pricing plans to suit different needs and budgets. Contact us for a personalized quote based on your specific requirements.



