Table of Contents
ToggleChange with confidence,
comply with ease
Streamlines the entire change lifecycle, providing
a centralized platform for managing all system modifications
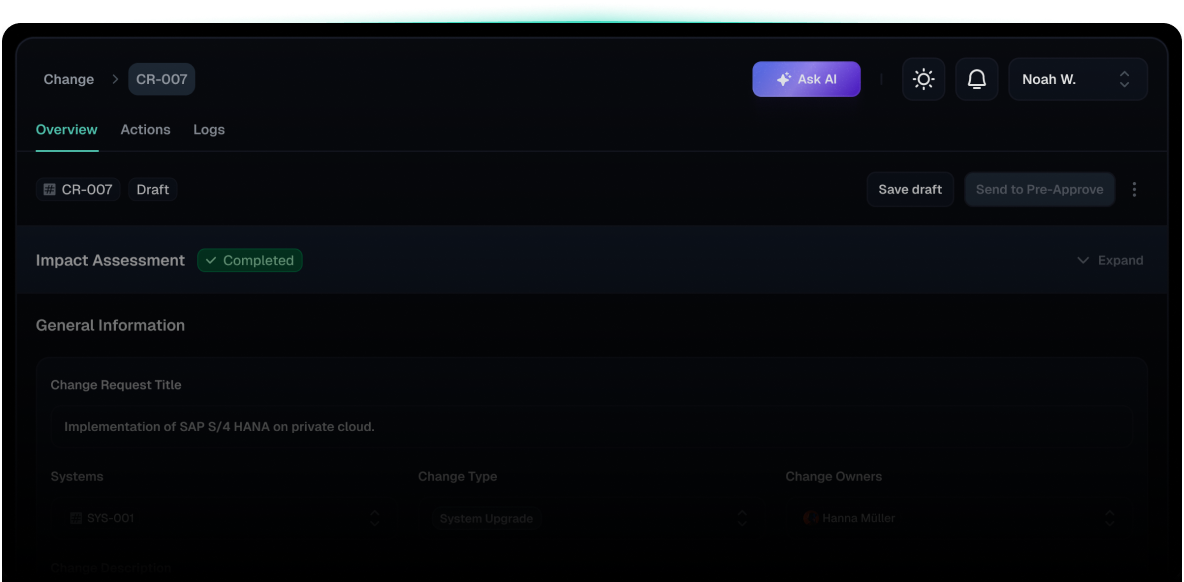
Change Module
Users can initiate new change requests with customizable unique IDs, categorized by change type, affected systems, and impact assessments.
Categorized change requests with unique IDs.
Fully traceable approval workflows with real-time visibility
Seamless integration for synchronized document revisions
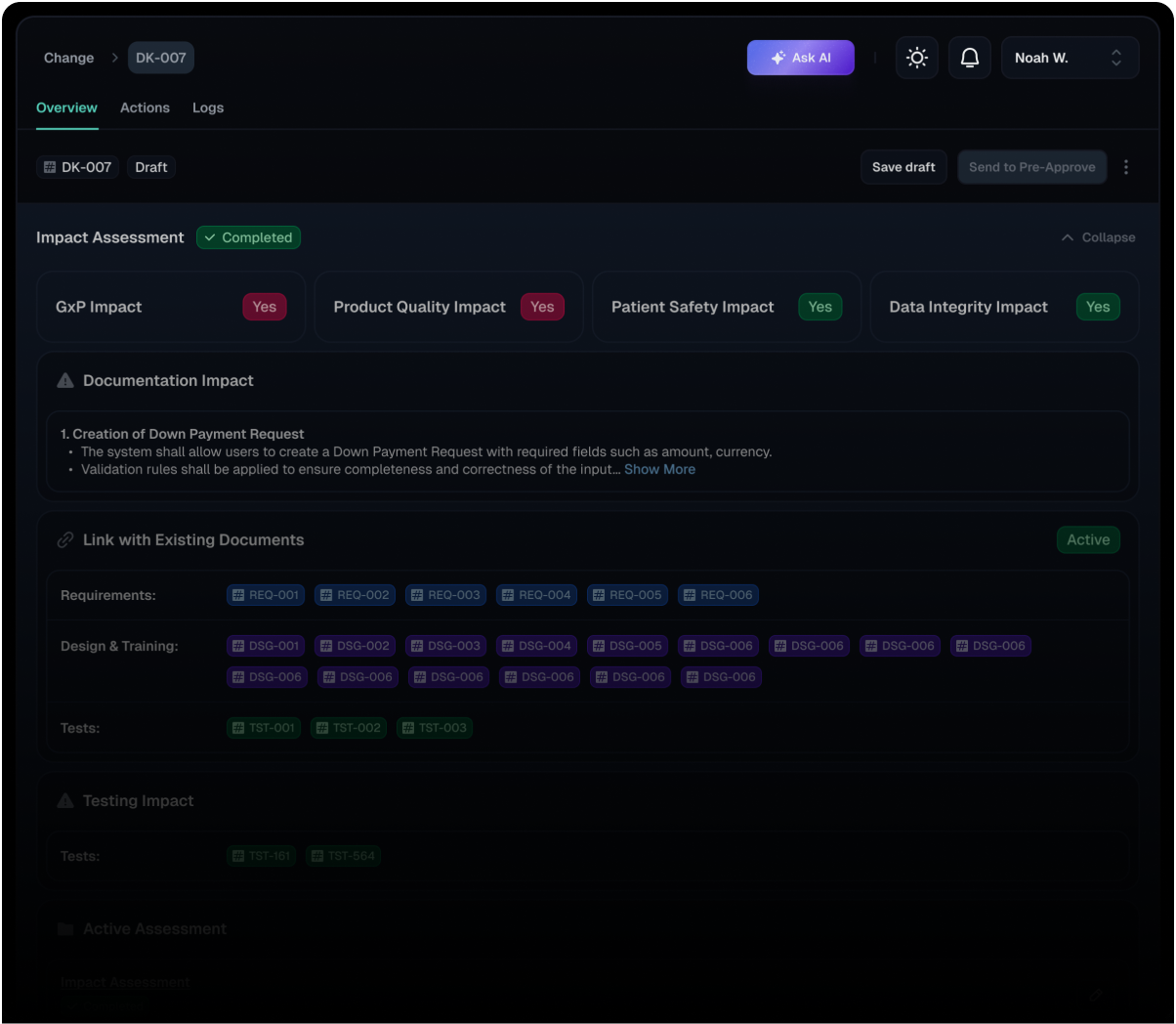
Seamless Integration
Seamlessly integrates Change Module with all related documentation, including requirements, designs, and tests. All updates to linked items ensure data integrity and facilitate comprehensive impact analysis throughout the change lifecycle.
Comprehensive Impact Assessment
Provides a structured framework for conducting thorough impact assessments, enabling organizations to proactively identify and mitigate potential risks associated with proposed changes. Evaluate effects on GxP, product quality, patient safety, data integrity, and documentation to facilitate informed decision-making and ensure compliance throughout the change lifecycle.
All Products
Change Management
Centralized tracking with real-time updates and compliance.
Deviation Management
Deviation tracking with structured actions and compliance.
Test Management
Efficient test management with real-time tracking and traceability.
Periodic Review Management
Automated reviews for compliance and integrity.
Smarter insights
bigger impact.
Computerized system validation is the backbone of safe,..
- Life Science, Validation Management
Data Integrity Policy for Pharmaceutical Industry is a set..
- Lifecycle, Validation Management
ALCOA principles are the five pillars, Attributable, Legible, Contemporaneous,..
- Life Science, Quality Assurance
In this blog, we cover: What Are Data Integrity..
- Life Science, Lifecycle
Challenges in maintaining data integrity is important for reliable..
- Quality Assurance, validfor
Computerized system validation is the process of proving that..
- Lifecycle, Validation Management
Frequently Asked Questions
How does Validfor's Change Module streamline the change control process?
Can I customize the change request workflow in Validfor?
Yes, Validfor allows you to customize the change request workflow to match your organization’s specific needs. You can define different approval levels, assign roles and responsibilities.
How does Validfor ensure traceability in change management?
Validfor ensures traceability by automatically linking change requests to related documents, such as requirements, designs, and tests. Any changes made to these linked documents are automatically reflected in the change request, providing complete visibility and control over the impact of changes.
How does Validfor handle emergency changes?
Validfor allows you to define and manage emergency change requests with expedited workflows and approvals, ensuring that critical changes are implemented quickly and safely.
Can I track the history of changes made to a system in Validfor?
Yes, Validfor maintains a complete history of all changes made to a system, including who made the change, what was changed, and when it was changed. This provides full transparency and auditability.
How does Validfor support impact assessment for changes?
Validfor provides a structured framework for conducting impact assessments, allowing you to evaluate the potential impact of changes on various aspects, such as GxP, product quality, and patient safety.
How does Validfor support electronic signatures for change requests?
Validfor integrates electronic signature capabilities, allowing authorized users to electronically sign change requests and related documents, ensuring compliance with 21 CFR Part 11 requirements.
Can I generate reports on change management activities in Validfor?
Yes, Validfor provides customizable reports on change management activities, allowing you to track key metrics, identify trends, and demonstrate compliance.






