Table of Contents
ToggleFrom Detection to Resolution
Control Every Deviation
Streamlines the entire change lifecycle, providing
a centralized platform for managing all system modifications
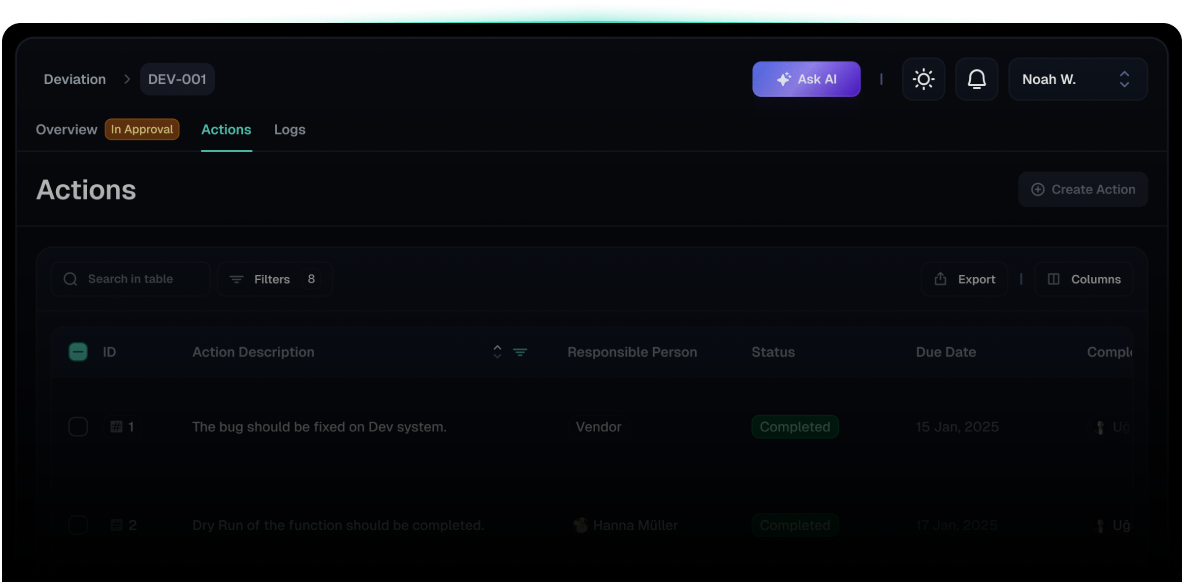
Deviation Module
Capture and categorize all validation-related deviations
and link them directly to their source tests or activities.
Manage quality events with impact levels and guided actions.
Ensure compliance with a structured approval workflow.
Track all progress through real-time dashboards.
Flawless Integration
Offers a sophisticated, integrated approach to resolving deviations identified during the testing process.
By seamlessly linking with Test Module functionalities, the platform provides immediate access to comprehensive test execution data, including specific steps, results, and associated documentation.
This facilitates thorough investigation, root cause analysis, and informed decision-making for effective deviation resolution ensuring comprehensive documentation and adherence to regulatory requirements throughout the validation lifecycle.
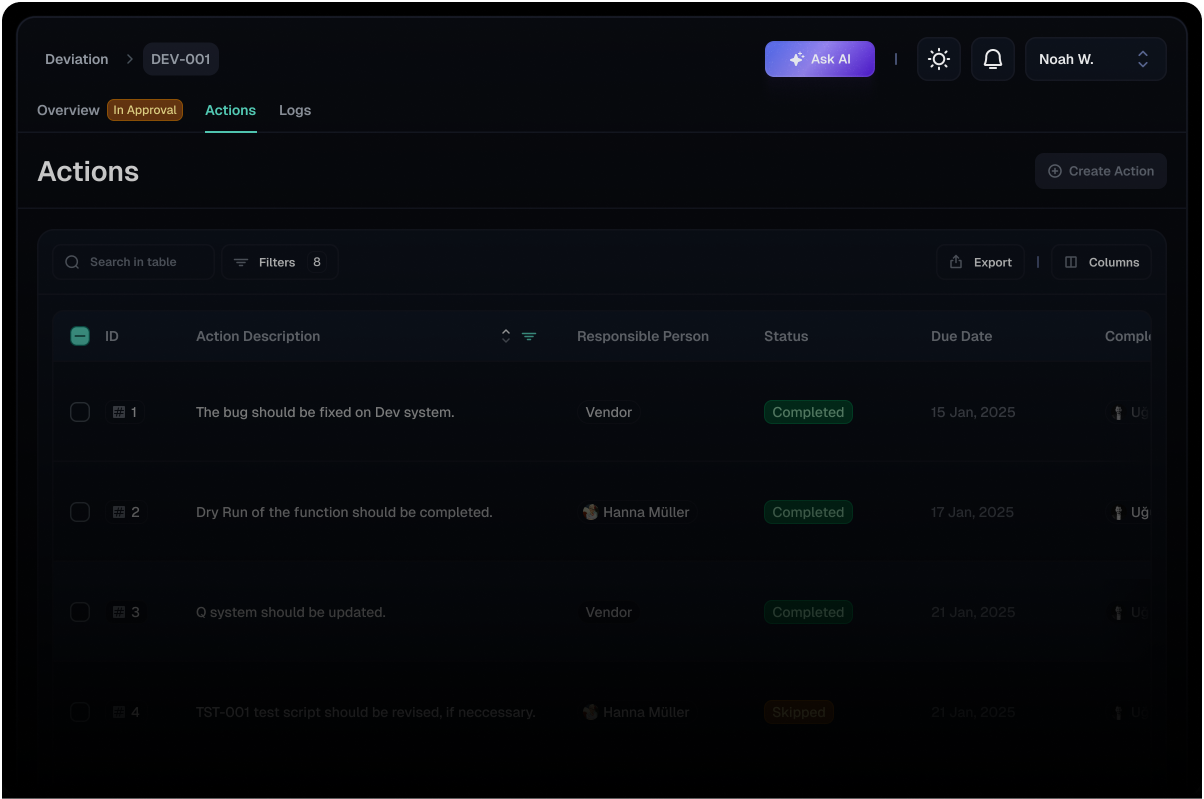
Structured Action Tracking
Provides a structured framework for tracking and managing corrective and preventive actions (CAPA) associated with deviations.
Assign responsibilities, set target completion dates, and monitor progress with ease, ensuring timely resolution and facilitating continuous improvement.
This structured approach promotes accountability and enhances the efficiency of deviation management workflows.
All Products
Change Management
Centralized tracking with real-time updates and compliance.
Deviation Management
Deviation tracking with structured actions and compliance.
Test Management
Efficient test management with real-time tracking and traceability.
Periodic Review Management
Automated reviews for compliance and integrity.
Smarter insights
bigger impact.
Computerized system validation is the backbone of safe,..
- Life Science, Validation Management
Data Integrity Policy for Pharmaceutical Industry is a set..
- Lifecycle, Validation Management
ALCOA principles are the five pillars, Attributable, Legible, Contemporaneous,..
- Life Science, Quality Assurance
In this blog, we cover: What Are Data Integrity..
- Life Science, Lifecycle
Challenges in maintaining data integrity is important for reliable..
- Quality Assurance, validfor
Computerized system validation is the process of proving that..
- Lifecycle, Validation Management
- See all articles
Frequently Asked Questions
How does Validfor help manage deviations, deficiencies, and defects?
Validfor provides a centralized platform to track, investigate, and resolve deviations, deficiencies, and defects identified during validation activities. You can categorize quality events, assign responsibilities, track corrective actions, and monitor progress towards resolution.
Can I link deviations to specific tests or validation activities in Validfor?
Yes, Validfor allows you to link deviations directly to the source tests or validation activities where they were discovered. This provides valuable context for investigation and root cause analysis.
How does Validfor ensure compliance in deviation management?
Validfor ensures compliance by enforcing a structured approval workflow for deviations, capturing electronic signatures, and maintaining detailed audit trails. This provides evidence of compliance with regulatory requirements.
How does Validfor facilitate collaboration in deviation management?
Validfor allows you to assign responsibilities, share information, and track progress on deviations, fostering collaboration among team members and stakeholders.
Can I define different categories and severities for deviations in Validfor?
Yes, Validfor allows you to define different categories and severities for deviations, enabling you to prioritize and manage them effectively.
How does Validfor support the escalation of deviations?
Validfor allows you to define escalation paths for deviations, ensuring that critical deviations are brought to the attention of the appropriate personnel for timely resolution.






